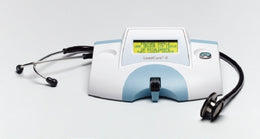
BD Respiratory Test Kit BD Veritor™ System Antigen Detection SARS-CoV-2 / Influenza A + B Nasal Swab Sample 2 X 30 Tests - KT/1 - 256095-KT
Our Policies
Privacy policy
Payment policy
Shipping policy
Regular price
$2,288.99
-Liquid error (snippets/product-price line 34): Computation results in '-Infinity'%
Regular price
$2,288.99
Certain items may require either a prescription, medical license, or a business facility license to process. If your product falls in that category, we will reach out to you to obtain proper documentation.
If you already have a prescription, you may email us at support@healthcareorigin.com along with the order number.
- Description
- NEW ITEM - Shipments expected to begin around Sept 1, 2021
- BD Triplex assay requires Veritor™ System firmware version 5.5 or greater; firmware version info is displayed on screen when turning on the analyzer or on the orange sticker if it is attached to the analyzer
- BD Veritor Plus System SARS/Flu (Triplex) Combo Promotional Kit - 2 X (256088) SARS-CoV-2/Influenza A + B Rapid Test Kit and (256066) BD Veritor Plus Analyzer
- BD Veritor System for Rapid Detection of SARS-CoV-2 and Flu A+B is For use under an Emergency Use Authorization only: https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-antigen-diagnostic-tests-sars-cov-2
- This test is authorized for use at the Point of Care (POC), i.e., i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation
- Laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, that meet the requirements to perform moderate, high, or waived complexity tests
- This product has been authorized only for the detection of proteins from SARS-CoV-2, influenza A and influenza B, not for any other viruses or pathogens
- The test is intended to be used with direct nasal swabs and is not validated for use with swabs in viral transport media
- Kit configured for testing anterior nasal swab samples freshly collected, processed, and dispensed directly onto assay test device
- Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status
- Positive results do not rule out bacterial infection or co-infection with other viruses
- Laboratories within the United States and its territories are required to report all SARS-CoV-2 results to the appropriate public health authorities
- Negative results should be treated as presumptive, do not rule out either Influenza or SARS-CoV-2, and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions
- Negative results should be considered in the context of a patient’s recent exposures, history and the presence of clinical signs and symptoms consistent with one of these infections
- Negative results for SARS-CoV-2 should be confirmed with a molecular assay, if necessary, for patient management
- The BD Veritor System for Rapid Detection of SARS-CoV-2 & Flu A+B is intended for use in point of care settings by trained healthcare professionals or other users specifically instructed in the use of BD Veritor Systems and proper infection control procedures
- Time to result: 15 minutes, test device can be read at 15 minutes but no later than 20 minute
- Each test kit contains: (30) Single Use Test Devices, (30) Single Use Reaction Tubes with 400 µL Reagent, (30) Sterile Single Use Sampling Swabs, 1 Positive SARS-CoV-2 Control Swab, 1 Positive Flu A Control Swab, 1 Positive Flu B Control Swab, (3) Paperboard Tube Stands, IFU, Quick Ref Card, and Nasal Sampling Guide
COMBO KIT, SARS-COV-2/FLU A+B W/VERITOR D/S
| Item Id | 1193638 |
| MF Id# | 256095 |
| Brand | BD Veritor™ System |
| Manufacturer | BD |
| Country of Origin | Unknown |
| Application | Respiratory Test Kit |
| CLIA Classified | CLIA Waived |
| Contents 1 | (2) Test Kits SARS-CoV-2 / Influenza A + B (30 Test each Kit), BD Veritor™ Plus Analyzer |
| For Use With | For use with the BD Veritor™ Plus Analyzer running Firmware version 5.50 or late |
| Number of Tests | 2 X 30 Tests |
| Product Dating | McKesson Acceptable Dating: we will ship >= 90 days |
| Purchase Program Type | Promotion |
| Reading Type | Machine Read |
| Sample Type | Nasal Swab Sample |
| Specialty | Immunoassay |
| Test Format | Test Device Format |
| Test Method | Chromatographic Digital Immunoassay |
| Test Name | SARS-CoV-2 / Influenza A + B |
| Test Type | Antigen Detection |
| Time to Results | 15 Minute Results |
| UNSPSC Code | 41116205 |